In HVAC engineering, Enthalpy is the measurement of the total heat content in a parcel of air. Unlike a standard thermometer, which only measures Sensible Heat (temperature you can feel), enthalpy accounts for both sensible heat and Latent Heat (energy stored in water vapor). This distinction is critical because cooling humid air requires much more energy than cooling dry air at the same temperature. Think of enthalpy as the “Full Energy Load” that your air conditioner has to wrestle with.
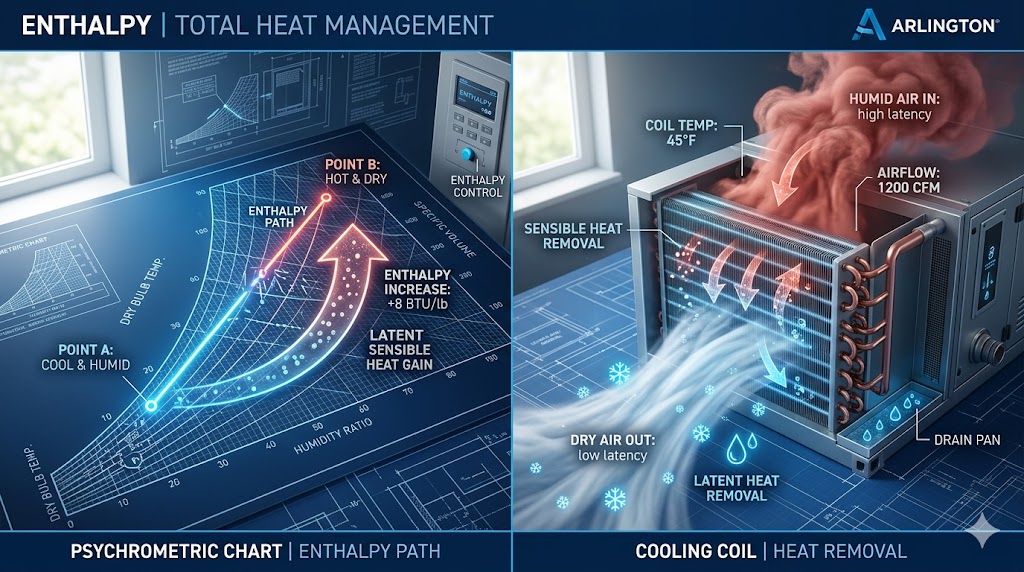
The concept of enthalpy is most important when discussing “Economizers” or free cooling. An economizer brings in outdoor air to cool a building when the outside temperature is low. However, if that cool outdoor air is extremely humid, its enthalpy might actually be higher than the indoor air you are trying to replace. In this scenario, your AC unit would have to work harder to remove the moisture than it would to simply recirculate and cool the indoor air. Understanding enthalpy is the difference between a system that is truly energy-efficient and one that is just “guessing” based on temperature alone.
To see the difference between what your thermometer says and the true energy load of your air, use the Enthalpy & Total Heat Auditor below.
What is enthalpy in HVAC systems
Enthalpy refers to the amount of internal energy inside the system together with the product of its volume and pressure. When it comes to enthalpy in HVAC systems, it is often assumed that this process is at constant pressure. This is the reason why a change in enthalpy will be equivalent to the heat released or absorbed.
How is enthalpy used in HVAC
Transferring heat, a form of energy, is the primary core function of HVAC systems. The Law of Conservation of Energy, the first law of thermodynamics, states that you cannot create or destroy energy. Instead, you can only transfer or change energy from one form to another.
Based on this, you can deduce that removing heat energy then transferring this somewhere, often outdoors, is the only way of cooling down a space. This is usually achieved through heat absorption from the airstream and distributing the cool air to the room you want to control.
You can think of conditioned air as something similar to a sponge that can soak heat up. The air that is now filled with unwanted heat will be routed back through a Return Air pathway for the process to start all over again.
This newly absorbed heat gets expelled and wrung out, with the cool air distributed once again in a constant cycle. The process of wringing out is primarily achieved through mechanical cooling like a compressor or free cooling courtesy of an air-side economizer.
💡 Pro Diagnostic Tip: The core reason enthalpy is used over simple temperature is to account for latent heat. When you need to strip moisture out of the air to prevent a “clammy” feel, you often use a Reheat system. Reheat allows the AC to lower the air’s enthalpy (removing moisture) and then add back just enough sensible heat for comfort.
Importance of enthalpy in HVAC
Mechanical cooling makes use of electricity for transferring the heat from the air stream to a different medium like refrigerant or water. Free cooling makes the most of favorable conditions outdoors to introduce air with greater capacity for heat absorption instead of the air being brought back to the system from the area you want to control.
Many economizes look only at the Dry Bulb temperature, or the reading being shown on the thermometer to identify the best option to use. What makes enthalpy very important for making this determination is the fact that outdoor air is a combination of dry air and water vapor.
This means that moist air’s enthalpy includes the heat absorption capacity of dry air known as sensible heat and the capacity of evaporating water in the air known as latent heat. The total enthalpy or the combination of latent and sensible heat values is used to carry out calculations to the cooling and heating processes. It is because the amount of moisture or the Relative Humidity of a certain parcel of air that is expressed as a percentage of the full moisture capacity can change the amount of energy that the air can transfer.
The main idea of free cooling is saving energy. This is why it will help to determine that the energy that a cooling coil uses is proportional to the difference in the enthalpy between air before this gets inside the coil and the air once it exits the coil.
💡 Pro Diagnostic Tip: In commercial buildings, managing enthalpy is the primary job of a TFA (Treated Fresh Air) unit. These units use enthalpy wheels to swap both heat and moisture between incoming and outgoing air, ensuring that fresh air doesn’t spike your building’s total energy load.
Factors that help determine enthalpy
To identify if free cooling will be the most energy-efficient option, you need to look at the air that enters the coil or the Dew Point of Return Air. Dew Point refers to the temperature at which Relative Humidity gets to 100% and starts condensing to water droplets or called Dehumidification.
When the Dew Point of entering air gets higher than the preferred air that leaves the coil or the Supply Air temperature, dehumidification will take place and result in the so-called Wet Coil. During instances of Wet Coil, the most energy-efficient choice is cooling the airstream with the lowest enthalpy. It might or might not be the airstream with the least Dry Bulb temperature because the levels of moisture have an important role played in determining enthalpy.
When the enthalpy of return air is higher compared to the outside air, cooling it down will require more damage. This is even when the temperature of outside air dry bulb temperature happens to be higher compared to the return air dry-bulb temperature.
When the return air dew point is lower compared to the supply air setpoint, dehumidification won’t take place and this leads to a dry coil situation. It is a different situation since the humidity ratios are similar for both leaving and entering air streams. The most efficient method for dry coils is cooling the airstream with the least dry bulb temperature, whatever the comparative humidity levels might be.
These calculations are all done with relative humidity and dry bulb temperature levels. However, the different applications of the two variables can lead to various factors that must be considered when trying to determine the option that will be the most energy-efficient method of cooling at the time.
Ideally, both the outside air and the return air must be constantly evaluated to make sure that you will make get the best out of the most effective method. All of these factors go into determining the enthalpy in HVAC systems.
How to calculate enthalpy in HVAC
Enthalpy in HVAC is easy. It is merely a state function that is only dependent on the current state of equilibrium identified by the volume, pressure and internal energy of the system. Enthalpy is an extensive quantity. Measurement of enthalpy in HVAC is used to determine the total heat change in fluid, whether it is water, refrigerant, or air.
Here are the steps to calculate enthalpy in HVAC:
- Start by calculating the air enthalpy. Multiply the air temperature in degrees Celsius by 1.007.
- Subtract 0.026 from the product.
- Calculate the water vapor enthalpy. Multiply the air’s water content and the air temperature.
Add the water vapor enthalpy and the air enthalpy to know the overall atmospheric enthalpy.